Pharmaceutical Systems Integration in Jiuduhe, Beijing
We provide specialized pharmaceutical systems integration focusing on sterile filling line automation, cleanroom batch controls, and unit-level traceability. Our engineering teams design and deploy deterministic PLC logic, high-fidelity historian systems, and validated SCADA platforms to ensure absolute data integrity and 21 CFR Part 11 compliance across high-stakes manufacturing and packaging environments.
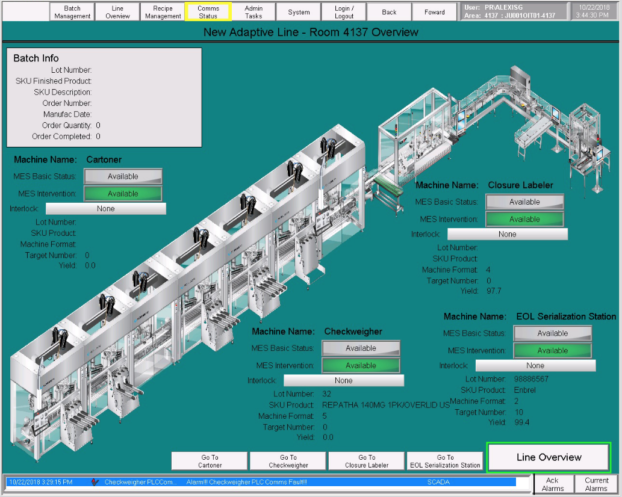
Modern pharmaceutical infrastructure relies on the precise orchestration of automated mechanical processes and immutable data management. Integration in this vertical involves the complex coordination of sterile filling lines, environmental monitoring systems, and multi-stage batch control logic. Our engineering focus centers on stabilizing the technical backbone of these regulated environments, ensuring that every control loop is deterministic and every state change is logged for auditable traceability. This involves hardening industrial networks and implementing secure telemetry to protect process variables from sterile production rooms to automated packaging stations. We architect integrated control layers that facilitate real-time communication between field instruments and enterprise historian systems. By implementing high-performance HMI dashboards and ISA-88 batch standards, we enable manufacturers to achieve consistent product quality while reducing the risk of batch losses. This technical rigor ensures that critical manufacturing environments satisfy rigorous validation protocols and maintain operational uptime in cleanroom manufacturing suites and quality laboratories.
Providing technical integration services to industrial facilities within the Jiuduhe metropolitan area and throughout Beijing.
Technical content for Pharmaceutical Systems Integration in Jiuduhe, Beijing last validated on April 5, 2026.
Services
Sterile Filling Line Integration
Synchronization of high-speed filling, capping, and sealing logic slaved to master controllers for precision dosing and 100% assembly verification.
Cleanroom Environmental Monitoring
Integration of precision HVAC logic and pressure differential monitoring to maintain ISO-standard sterile production environments and personnel safety.
Automated Batch Control Systems
Design of ISA-88 compliant logic frameworks for pharmaceutical recipe management, ingredient weighing, and automated reaction control.
Unit-Level Traceability Telemetry
Architecture of high-bandwidth data acquisition networks slaved to edge logging hardware for unit-level tracking and serialized data integrity.
Pharmaceutical SCADA & Historian
Architecture of high-performance visualization systems slaved to validated databases for real-time process monitoring and 21 CFR Part 11 auditability.
Packaging & Serialization Automation
Engineering of secondary packaging logic including high-speed labeling, case packing, and integrated serialization camera systems for compliance.
Our Process
Requirement Audit & Validation Plan
Identification of specific I/O magnitude, GAMP 5 classification, and required validation protocols to define the baseline engineering scope.
Logic Architecture & Validation Design
Engineering of deterministic control sequences slaved to functional specifications to ensure logic integrity and repeatability for regulated production.
System Implementation & Integration
Physical installation of control hardware and field instruments combined with logical pairing via hardened industrial protocols and secure OT segments.
Operational Validation & SAT
Execution of rigorous site acceptance testing (SAT) to verify safety interlocks, alarm handling, and auditable electronic records before formal handoff.
Use Cases
Orchestration of a multi-stage liquid filling line slaved to a master controller to ensure precise dosing and automated cap-torque verification in a sterile production room.
Deployment of a facility-wide environmental monitoring system (EMS) to track and log pressure differentials and sterile air quality metrics in real time across multiple suites.
Integration of high-speed robotic cells slaved to machine vision to inspect, pick, and place vials into primary packaging at rates exceeding 200 units per minute.
Architecture of a validated process historian for an API manufacturing plant, aggregating 5,000 plus process tags into an immutable SQL database for batch reporting.
Engineering of a turnkey secondary packaging line featuring integrated serialization, weight checking, and automated rejection logic to ensure compliance.
Technical Capabilities
- Safety-rated PLCs in pharmaceutical environments utilize dual-processor architecture to ensure deterministic fail-safe states during hardware or logic faults.
- Industrial historians utilize data compression algorithms and store-and-forward technology to prevent data loss during communication interruptions in the OT network.
- Cleanroom pressure control logic utilizes high-fidelity PID loops slaved to millisecond-response dampers to prevent particulate contamination ingress.
- ISA-101 high-performance HMI standards utilize grayscale backgrounds and color-for-exception symbols to reduce operator fatigue and improve alarm response times.
- The implementation of electronic signatures and timestamped audit trails within SCADA environments is mandatory for satisfying 21 CFR Part 11 requirements.

Pharmaceutical Systems Integration support
Pharmaceutical Systems Integration services for facilities in Jiuduhe, Beijing, China .
Pharmaceutical Systems Integration support
Pharmaceutical Systems Integration services for facilities in Jiuduhe, Beijing, China .

Pharmaceutical Systems Integration support
Pharmaceutical Systems Integration services for facilities in Jiuduhe, Beijing, China .

Pharmaceutical Systems Integration support
Pharmaceutical Systems Integration services for facilities in Jiuduhe, Beijing, China .

Pharmaceutical Systems Integration support
Pharmaceutical Systems Integration services for facilities in Jiuduhe, Beijing, China .

Pharmaceutical Systems Integration support
Pharmaceutical Systems Integration services for facilities in Jiuduhe, Beijing, China .

Pharmaceutical Systems Integration support
Pharmaceutical Systems Integration services for facilities in Jiuduhe, Beijing, China .

Pharmaceutical Systems Integration support
Pharmaceutical Systems Integration services for facilities in Jiuduhe, Beijing, China .

Pharmaceutical Systems Integration support
Pharmaceutical Systems Integration services for facilities in Jiuduhe, Beijing, China .
Frequently Asked Questions
How is logic validation managed in regulated pharmaceutical environments?
We follow GAMP 5 methodologies, providing comprehensive documentation including IQ and OQ protocols to verify that all control logic performs according to the design specification.
Can legacy filling lines be integrated into modern historian systems?
Yes, we utilize protocol converters and distributed I/O blocks to extract data from older instrumentation and normalize it for centralized SCADA monitoring and historian logging.
What is the importance of ISA-88 standards in batch control?
ISA-88 provides a modular framework for defining batch processes, separating equipment control logic from recipe management to ensure consistency and easier validation.
How do you handle data integrity during network outages?
We implement edge devices with store-and-forward capabilities that buffer process data locally and automatically backfill the primary historian once the connection is restored.
Validate Your Pharmaceutical Infrastructure
Submit technical requirements for an engineering review of your pharmaceutical production line, cleanroom monitoring, or batch control project.
Request Engineering Audit